Understanding the lived experience of follicular lymphoma (FL) is essential to improving care, research, and support. While clinical data tells us how treatments work, patient insights tell us how people actually live with this disease.
In October 2025, we conducted the FLF’s annual Global Patient Survey to better understand the challenges, concerns, and priorities of people living with follicular lymphoma. More than 1,000 people took part from over 50 countries worldwide – sharing their experiences across different stages of the disease and from healthcare systems around the world. These insights offer an important perspective on where support is working well and where further progress is needed.
Follicular lymphoma is typically a long-term condition, and experiences can change significantly over time. People may move between periods of monitoring, treatment, remission, and relapse, often with long gaps between major clinical milestones.
We ran this survey to capture those experiences more clearly and consistently. Our aim was to understand what patients find most challenging, what information they feel is missing, and how their needs evolve throughout their journey.
By collecting this data directly from patients, we can ensure that patient perspectives help shape research priorities, educational resources, and advocacy efforts. The survey also allows us to identify shared experiences across regions, while recognising that access to care and support can differ widely.
The survey was co-designed with members of the FLF’s dedicated patient involvement group, known as the Super Supporters. They helped shape the questions, language and focus of the survey, ensuring it reflected real patient priorities and concerns.
We then shared the survey over 3 weeks, in English and Spanish across all our patient networks online: social media channels, our website, newsletters, and word of mouth within the global FL community. This approach helped us reach people of all ages, any stage of the disease and across a wide range of countries.
After the survey closed, we explored the findings in more depth through two follow up focus groups with members of the Super Supporters. One session focused on understanding the nuances behind some of the key results, while the second focused specifically on challenges around understanding and accessing clinical trials. During this second workshop, we also worked together to begin co-creating potential solutions to address the barriers patients described.
We have shared what people told us in the survey and focus groups with doctors, researchers, and others working in follicular lymphoma. By bringing real patient experiences to clinical, research, and industry discussions, we aim to raise awareness, improve understanding, and encourage change. These insights help guide future research and care, so that new treatments and cures are shaped by what truly matters to people with FL and those who support them.
Over the course of 3 weeks during October 2025, a total of 1,042 people responded to the FLF’s 2025 Global Patient Survey. Participants came from 52 countries, reflecting a broad international reach. The majority of respondents (71%) were from the USA, UK and Spain; then 7-8% each were from other European countries, Latin America, and Australia; and 5% from Canada.
The survey was available in both English and Spanish languages, which helped reach people from different linguistic backgrounds and healthcare settings.
Respondents represented all ages and stages of follicular lymphoma. This included people who were recently diagnosed right through to those living with FL for 10 years or more, those who had not yet required treatment, individuals who had received one or more lines of therapy, and people living with stable disease, remission, or relapse. This diversity of experiences provides confidence that the insights we are highlighting are truly representative of the wider FL community.


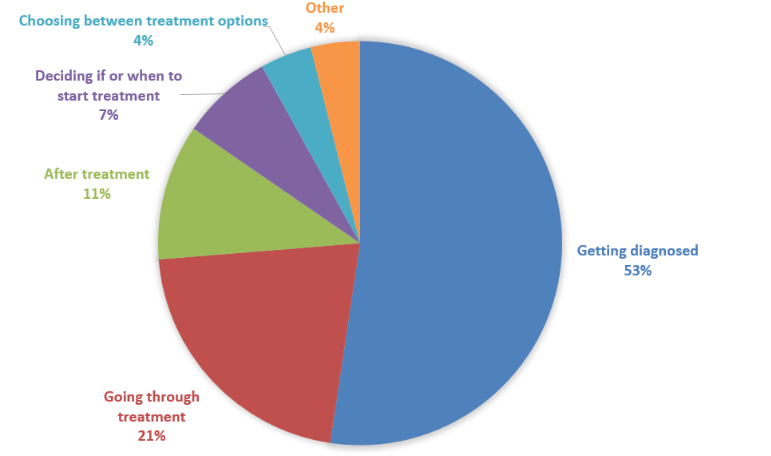
Survey responses showed that the most difficult aspects of living with FL vary, but several common themes emerged. For most, the point of diagnosis was reported as the hardest moment (50%). The initial uncertainty, combined with limited understanding of what the diagnosis means in the long term, can be particularly challenging.
Others identified later stages as more difficult, including going through treatment (20%), after treatment (10%), starting treatment (7%), or choosing between treatments (4%). These moments often bring new decisions and renewed concerns.
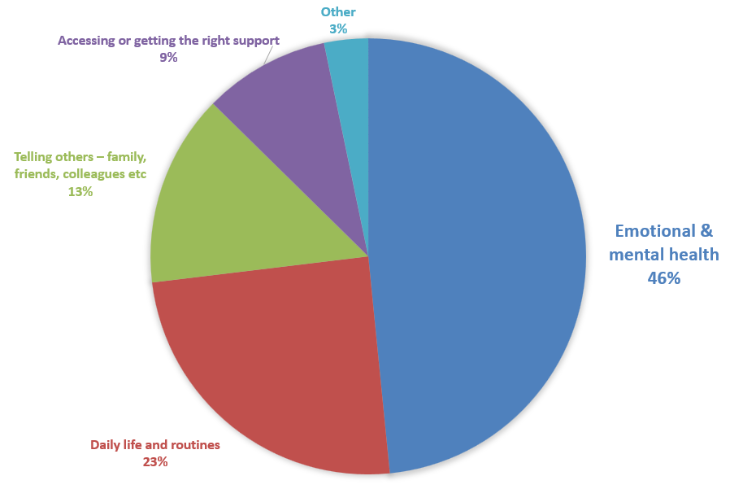
When asked about ongoing challenges, he heard loud and clear that emotional and mental health is the hardest challenge for all respondents (46%). The next biggest ongoing challenges for FL patients were daily life and routines (23%), telling others (13%), and accessing the right support (9%). During our virtual roundtable discussion with the FLF’s Super Supporters, many described long term anxiety, fear of progression, or uncertainty about the future, even during periods when the disease was stable. The mental health burden of watch and wait made many relieved to start treatment, only to find out about long term side effects further down the line.
At the same time, some people reported that they feel they are coping well, particularly as they gain experience and knowledge over time. Put together, this highlights the importance of understanding fully what potential long term side effects of treatment are, the impact on treatment sequencing for people to make informed choices, and the need for flexible support throughout.
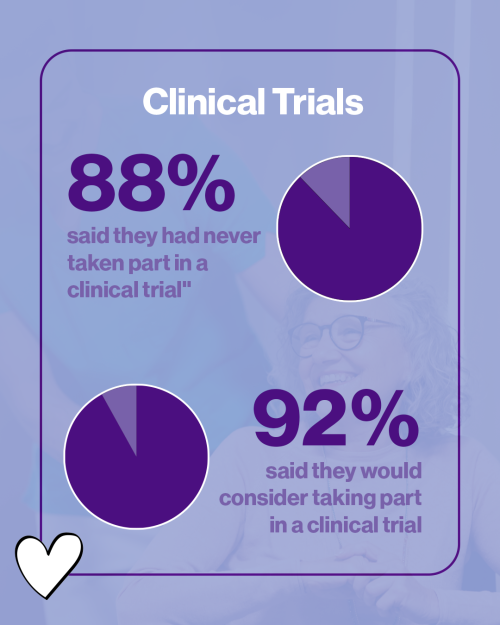
Clinical trials play a key role in improving treatments for follicular lymphoma, but participation remains limited. Most respondents reported that they have never taken part in a clinical trial (88%). However, the survey also showed a clear interest in learning more about trials and a willingness among some patients to consider participation in the future. Only 5% of respondents said they would not want to participate in a clinical trial if offered – with a large proportion saying they would (42%) or were not sure (50%).
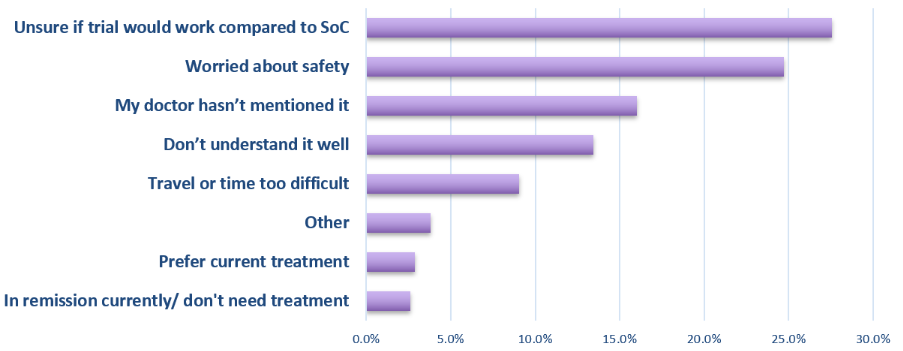
Respondents told us about several barriers they face in joining a clinical trial. Many felt unsure about how helpful (28%) or safe (25%) clinical trials might be. Many said trials were never discussed with them as an option (16%), while others found it hard to get clear, reliable information (13%). Practical barriers, such as trials not being available close to home were another important factor (9%).
Through our subsequent co-creation workshop with FLF’s Super Supporters, we learned there is a huge need for general information around clinical trials, to address misconceptions. Understandably if there is any confusion, patients will just say no. While interest exists, better communication and more accessible information are needed to support informed decision making around clinical trials.
Further supporting the earlier finding that the mental health burden of FL is generally widespread – making a decision around participating in clinical trials only adds to this. Offering mental health support with the decision would ensure informed choices, build trust and even incentivise some patients to join a trial and contribute to future breakthroughs in curing FL.
The findings from this survey reinforce the how crucial it is to including patient voices in shaping the future of follicular lymphoma care.
The results highlight shared challenges that many people living with FL experience, regardless of where they live or how long they have been diagnosed. They also point to clear opportunities to improve information, communication, and support across the patient journey.
We will use these insights to guide future patient resources, inform research discussions, and strengthen advocacy efforts. Future surveys will continue to build on this work, helping ensure that patient experiences remain central to progress in follicular lymphoma.
By taking part, you directly contribute to a growing evidence base that helps improve understanding, care, and outcomes for the wider community – scientific, clinical, industry, advocacy and more.
This survey was supported by sponsorship from Novartis, Genmab and AbbVie.
All of the above have had no influence on, control of, nor input into the development or performance of the survey.
Disclaimer: The information provided in this blog post is for educational purposes only and should not be considered a substitute for professional medical advice. Always consult with your healthcare provider for personalised guidance and treatment options.
Published: 30th March 2026